Description
AgX100® is intended to treat prostate cancer, intra-ocular malignancies, and other tumor types with known radiosensitivity
Based on the industry standard
Time-tested clinical experience
- Substantially equivalent in physical and dosimetric characteristics to the I-125 industry benchmark1
- Recognized as the most widely published and clinically proven I-125 seed for the treatment of localized prostate cancer1
- Available in a wide range of activities
- 59.43-day half-life; 27-35 keV principle energy
Unparalleled quality
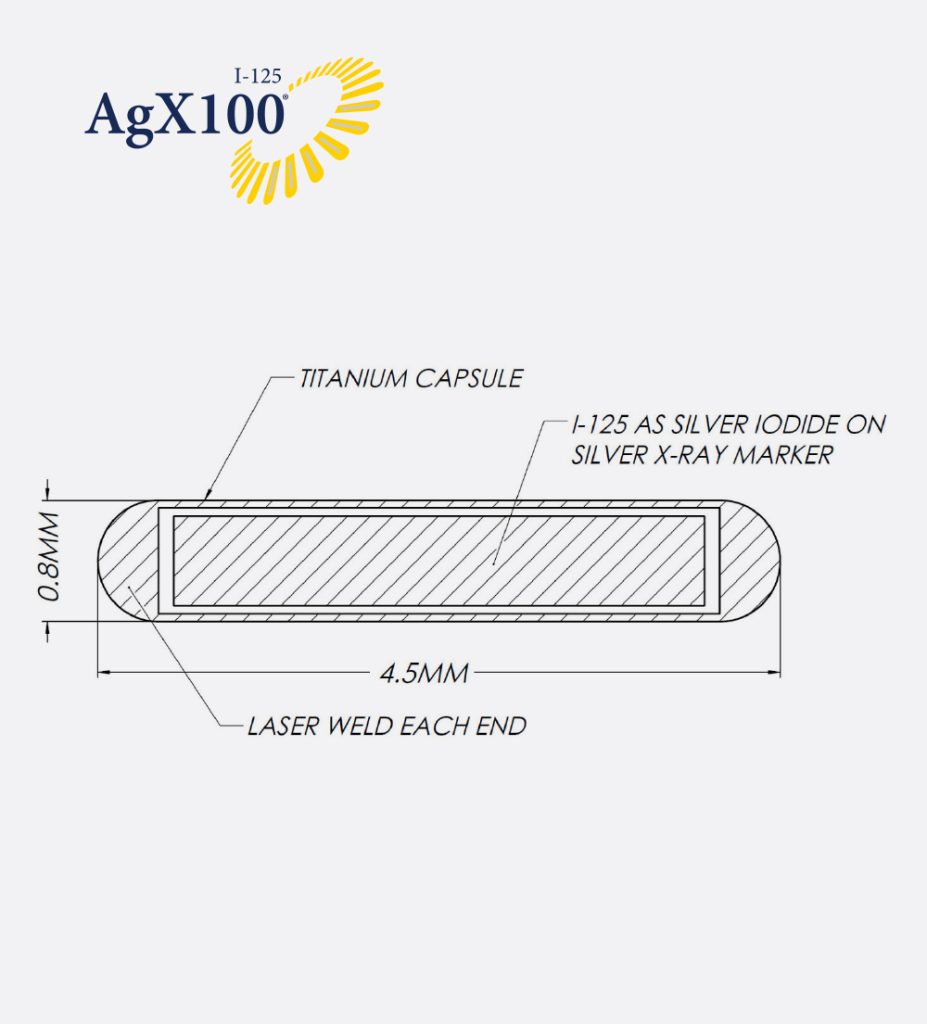
- Designed with a stable silver core for optimal imaging clarity
- Laser-welded titanium capsule
- I-125 sourced and manufactured in North America for reliable supply
I-125 BT has been widely used in the treatment of various tumors due to its compelling efficacy and potential for more general applicability.2

Interested in learning more?
To find out how to integrate AgX100® into your practice, speak with one of our brachytherapy experts.

- Data on file
- Wei S, Li C, Li M, et al. Radioactive Iodine-125 in Tumor Therapy: Advances and Future Directions. Front Oncol. 2021;11:717180. Published 2021 Sep 30. doi:10.3389/fonc.2021.717180
AgX100® I-125 source and accessories should be used by physicians who are qualified by training and experience in the safe use and handling of radionuclide brachytherapy sources and whose experience and training has been approved by the appropriate government authorities authorized to license the use of radioactive materials.
Information intended for the USA. Indications may vary in different countries; please either consult the appropriate IFU or your local distributor.


